Clinical Electronic Data Capture
Trialomics built and managed a scalable, compliant clinical data platform that enabled remote patient monitoring and connected patients, trial coordinators, sponsors, clinicians, and FDA-facing study teams through a secure digital workflow.
Scalable Clinical Data Platform for Remote Patient Monitoring
Trialomics partnered with Sana Health to support clinical trials for a novel wearable mask designed to help users reduce pain, improve sleep, and elevate mood. The product required more than a patient-facing app — it required a secure, validated platform that could collect data remotely and make that data usable across the full clinical trial ecosystem.
We built and managed the complete technology stack: mobile applications, Bluetooth device integration, secure cloud infrastructure, data capture workflows, validation documentation, and operational controls. The platform enabled patients to participate remotely while giving trial coordinators, sponsors, clinicians, and FDA-facing teams access to the information and oversight needed to run compliant studies.
We also hosted and operated the system under our cloud platform, Picard, providing end-to-end support for remote monitoring, data integrity, compliance evidence, and study lifecycle management.
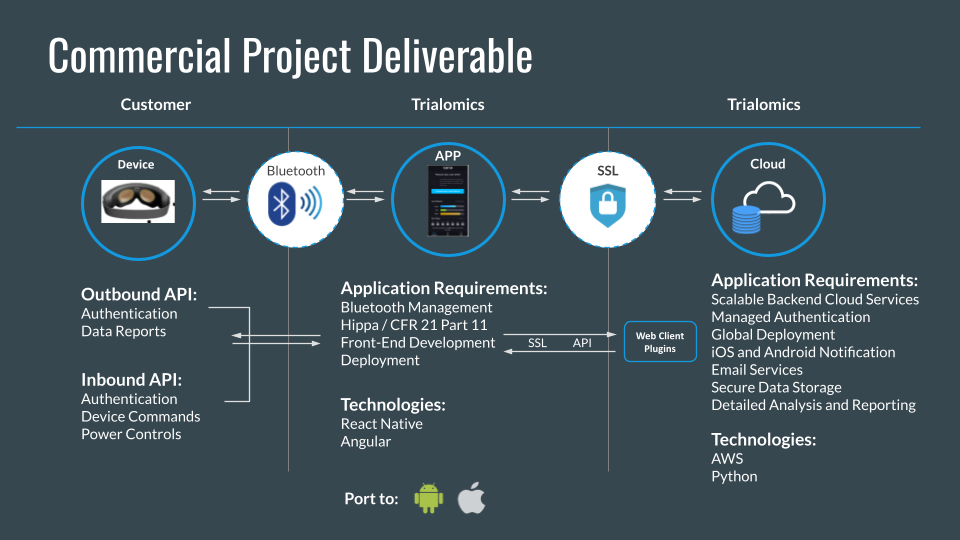
The diagram below illustrates how the wearable device, mobile applications, and cloud infrastructure connected patients with the operational and clinical stakeholders responsible for the trial.
What We Delivered
Trialomics delivered the platform, documentation, compliance operations, and technical management needed to support regulated remote patient monitoring at clinical trial scale.
Patient-Facing Applications
- • Mobile app development – Built clinical-grade Android and iOS applications for patient participation and remote data capture.
- • Device integration – Connected early Bluetooth prototypes to the mobile workflow so study data could be captured from the wearable device.
- • Participant experience – Designed user flows that supported study adherence, data capture, and remote monitoring outside the clinic.
Validation, Traceability & Data Integrity
- • System validation – Created validation documentation and testing strategy for the platform.
- • Clinical workflow verification – Verified that app, database, and cloud workflows aligned to study requirements.
- • Requirement traceability – Mapped requirements to test cases and evidence to support regulatory review and audit readiness.
️ Scalable Compliance & Hosting
The Picard platform provided a secure, scalable cloud environment for regulated data collection, monitoring, and analysis.
- • Compliant cloud operations – Managed hosting, security controls, access control, encryption, and operational procedures.
- • Operational evidence – Maintained task ledgers, audit trails, and documentation to support compliance oversight.
- • Risk management – Supported risk analysis, mitigation planning, and ongoing platform governance.
Connected Clinical Trial Stakeholders
- • Patients – Participated remotely through mobile workflows connected to the wearable device.
- • Trial coordinators – Monitored participation, data completeness, and study operations.
- • Sponsors and clinicians – Accessed the structured data and reporting needed to evaluate outcomes.
- • FDA-facing teams – Used validation, traceability, and compliance documentation to support regulated trial oversight.
Why This Matters
Remote patient monitoring only works when the software, cloud infrastructure, compliance model, and clinical workflow operate as one system. Trialomics helped turn an early wearable product into a managed clinical platform that could support regulated data capture across real-world participants and trial stakeholders.
- • Built the patient-facing and operational technology stack
- • Managed scalable, compliant cloud hosting and platform operations
- • Connected patients, coordinators, sponsors, clinicians, and FDA-facing teams through shared clinical data workflows
- • Produced validation and traceability evidence needed for regulated clinical studies
A managed platform for regulated remote monitoring
Trialomics combined software development, cloud operations, compliance documentation, and clinical workflow design to help a customer run FDA-sponsored studies with connected, remotely monitored patient data.